The Culture of Organoids
- 22 hours ago
- 8 min read
Malleeka Suy

Planetoid, humanoid, organoid–these words sound like the makings of a great futuristic space odyssey. Perhaps so, but one of these sci-fi-sounding elements represents a rapidly advancing avenue of research that could address age-old challenges in neuroscience: Organoids. Creating and transplanting these man-made organs into humans is much more advanced than you might think, and they are illuminating a path towards a better understanding of brain functions, diseases, and cures.
What are Organoids?
Organoids are self-assembling mini-3D structures cultured, or grown, from stem cells, to bear striking structural, genetic, and functional similarities to their human counterparts. Organoid research began in the early 1900s, mostly involving the isolation of preexisting cells. In 1975, keratinocytes (skin cells that produce keratin), and fibroblasts (connective tissue cells), were successfully cultured together to grow human skin colonies (Rheinwatd and Green, 1975). This discovery advanced studies involving skin grafts, dermatological conditions, and drug testing, and would ultimately lead to the isolation and reconstruction of many other organs.
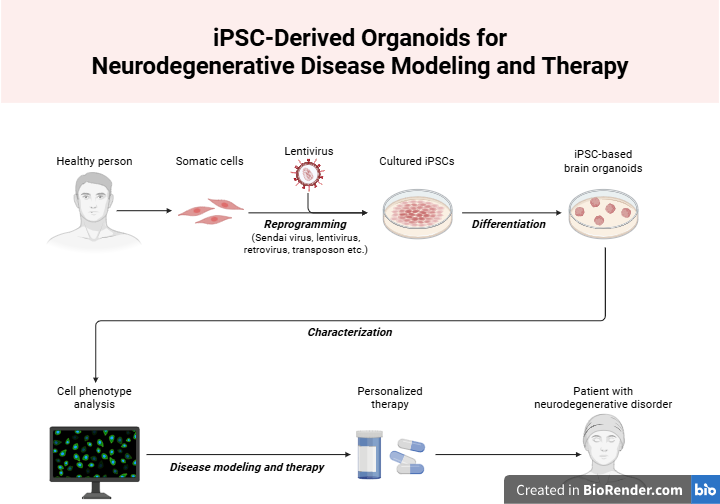
A significant landmark in organoid research was the discovery and isolation of stem cells. The most popular stem cells used for organoid culturing are embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs). ESCs are unspecialized cells collected directly from early-stage embryos, while iPSCs are unspecialized cells reprogrammed from specialized adult cells, such as skin or liver cells. You can think of unspecialized cells like a ‘blank slate’; given the appropriate growth conditions, researchers can develop them into any specialized cell and construct any organoid of their choice. In 2009, Toshiro Sato, then a postdoctoral researcher at the Hubrecht Institute in Utrecht, cultured what is widely considered the first organoid. Sato used single stem cells to form small intestines similar to those found in the human body, complete with villi protrusions for nutrient absorption and crypts, where new intestinal stem cells grow (Sato et al., 2009). Since then, organoid culturing protocols have been developed for all types of organoids, which have become excellent models in many biomedical applications, including regenerative research, disease modeling, and personalized medicine (Yang et al., 2023).
Brain Organoids
The first brain organoids, also termed cerebral organoids or cortical organoids, were developed in 2013 by developmental neurobiologist Madeline Lancaster, who developed a protocol for brain tissue culturing and growth as part of her post-doctorate work at the Institute of Molecular Biotechnology of the Austrian Academy of Science. Lancaster and other scientists started by isolating and culturing a class of embryonic cells that differentiate to form the nervous system. These cells make up a structure called the neuroectoderm and serve as the “building block” of the organoid Lancaster developed. The cells quickly grew in specialized environments that triggered developmental cues within the cells and supplied necessary nutrients for neural growth. By days 20-30, defined brain regions had formed (Lancaster et al., 2013). Staining experiments also confirmed that brain regions within the organoid grew in a similar manner to corresponding regions in human brains: Although the regions were distinct and separate, they still influenced the growth and development of one another (Lancaster et al., 2013). So far, cerebral organoids displayed similar patterns of distinct brain region development and growth interactions in comparison to human-developing brains. These findings marked an important step towards accurately modeling the human brain, as well as exploring the similarities and differences in human neurodevelopment in comparison to other species. As Lancaster exclaimed in an interview with Cell Stem Cell, “For the first time we can now ask, what makes our brains special or different?” (Lancaster, Interview with Cell Stem Cell, 2017).
The use of cerebral organoids also fills a large gap in using animals as models for studying human brain disorders. In the case of microcephaly, a neurological condition in which an infant’s head is much smaller than usual, mouse models with the disorder do not fully display the same traits as affected humans, making it challenging to analyze (Farcy et. al., 2022). Lancaster hoped that cerebral organoids could be used as a model for neurodevelopmental disorders by utilizing iPSCs from a patient with severe microcephaly to model the diseased brain. The resulting organoids displayed smaller neural tissues and few progenitor regions for neuron production–features consistent with the reduced brain size seen in microcephaly patients (Lancaster et al., 2013).
With further experimentation, Lancaster and her team also identified a possible cause of the smaller brain displayed by microcephaly patients. In cell division, a weblike spindle aligns to separate chromosomes. Lancaster noticed that in the division of radial glial cells, the primary progenitor cells for neurons, many of the spindles were vertical or slanted, as opposed to horizontal. This led to a faulty expansion of neural stem cells and the development of a smaller brain (Lancaster et al., 2013). This could also explain why mice do not display human-like microcephaly traits, as mice do not undergo the same level of neural cell expansion as humans do (Lancaster et. al., 2013). With Lancaster’s discoveries, brain organoids have become widely used in research on neurological disorders, including Alzheimer’s disease, Rett syndrome, and Zika virus (Zhang et al., 2024; Samsarasinghe et al., 2021; Esper et al., 2022).
Cortical organoids also hold significant promise for tackling brain injury response and treatment, a notorious challenge for researchers and clinicians. Scientists have managed to successfully transplant human brain organoids grown on culture dishes into mouse brains–complete with neuron differentiation, axon growth, and synaptic communication across the transplanted portions to the host (Mansour et al., 2018). This organoid grafting method is an invaluable tool in modeling disease within a living animal system, as well as neural circuit reconstruction. For example, a study from the Perelman School of Medicine at the University of Pennsylvania utilized organoid grafting to successfully integrate iPSC brain organoids into mice that suffered damage to the visual cortex, the part of the brain responsible for processing visual information. Not only did the grafts have a survival rate of 82% three months after being transplanted, but neurons also increasingly matured within these grafts and displayed similar signal firing rates compared to those from the host brain portions (Jgamadze et al., 2023). The visual cortex neurons of the organoid also responded with similar levels to host brain neurons during both exposure to on-and-off light stimulation and when light was shone in different positions (Jgamadze et al., 2023). The long-term survival rate, consistent neuronal activities, and adaptive responsiveness to visual stimuli suggested that brain organoids can successfully integrate and communicate with preexisting networks. These researchers are illuminating exciting avenues for brain circuitry function and response, with the possibility of neural regeneration on the horizon.
The Horizon for Organoids
A feat of biomolecular and cellular efforts, organoids have vastly advanced many fields of scientific research. However, much still needs to be improved within organoid formation itself. Because of their relatively short life span, most organoids, especially those derived from pluripotent stem cells, can only represent fetal tissue rather than mature adult tissue, leading to research skewed towards early-human brain development (Yang et al. 2023). Brain organoids also lack many crucial components and processes of a human brain, such as microglia (immune cells of the central nervous system), vascularization (the process of forming new blood vessels), and rely heavily on culture media for sufficient nutrient and oxygen supply (Urrestizala-Arenaza et al., 2024; Lancaster, Interview with Cell Stem Cell, 2017). This leads to an inner necrotic core in the brain organoid, or a “dead zone” of cells caused by limited oxygen and nutrient diffusion, making it challenging to form a fully accurate model of the brain. However, scientists have been on the move to address these problems. For example, researchers have found a way to coax the expression of genes that promote microglia development and vascularization, allowing functional maturation of the organoid and reducing the necrotic core (Cakir et al., 2019; Cakir et al., 2022). Blood vessel organoids could also be used as brain organoid vascularization to bring in nutrients to the brain via blood circulation–a twofold organoid technique (Wimmer et al., 2019).
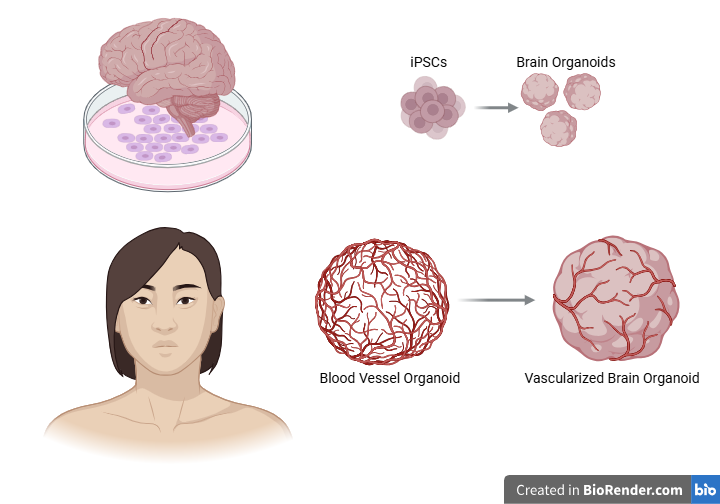
With fine-tuning these aspects comes a push and pull of progress and drawbacks, but it is well worth the time and investment. From understanding neurodevelopmental defects to treating traumatic injury, organoids have allowed for further ventures into subfields of neuroscience previously inaccessible. The prospects for organoid use also stretch beyond biological research, with applications extending into anthropology, engineering, and computer science. By culturing the perfect organoid, society is well on its way toward turning scientific hopes and dreams into reality.
References
Cakir, B., Tanaka, Y., Kiral, F. R., Xiang, Y., Dagliyan, O., Wang, J., Lee, M., Greaney, A. M., Yang, W. S., DuBoulay, C., Kural, M. H., Patterson, B., Zhong, M., Kim, J., Bai, Y., Min, W., Niklason, L. E., Patra, P., & Park, I. (2022). Expression of the transcription factor PU.1 induces the generation of microglia-like cells in human cortical organoids. Nature Communications, 13(1), 1-15. https://doi.org/10.1038/s41467-022-28043-y
Cakir, B., Xiang, Y., Tanaka, Y., Kural, M. H., Parent, M., Kang, Y., Chapeton, K., Patterson, B., Yuan, Y., He, C., Raredon, M. S., Dengelegi, J., Kim, K., Sun, P., Zhong, M., Lee, S., Patra, P., Hyder, F., Niklason, L. E., . . . Park, I. (2019). Engineering of human brain organoids with a functional vascular-like system. Nature Methods, 16(11), 1169-1175. https://doi.org/10.1038/s41592-019-0586-5
Cerchio, S., Cavaliere, F., & Magliaro, C. (2024). Limitations of human brain organoids to study neurodegenerative diseases: A manual to survive. Frontiers in Cellular Neuroscience, 18, 1419526. https://doi.org/10.3389/fncel.2024.1419526
Clevers, H., Lancaster, M., Takebe, T. (2017). Advances in Organoid Technology: Hans Clevers, Madeline Lancaster, and Takanori Takebe. Interview with Cell Stem Cell, 20(6), 759-762. https://doi.org/10.1016/j.stem.2017.05.014
Driehuis, E., Kretzschmar, K., & Clevers, H. (2020). Establishment of patient-derived cancer organoids for drug-screening applications. Nature Protocols, 15(10), 3380-3409. https://doi.org/10.1038/s41596-020-0379-4
Esper, N.B., Franco, A.R., Soder, R.B., Bomfim, R.C., Nunes, M.L., Radaelli, G., Esper, K.B., Kotoski, A., Pripp, W., Neto, F.K., Azambuja, L.S., Mathias, N.A., da Costa, D.I., Portuguez, M.W., da Costa, J.C. (2022). Zika virus congenital microcephaly severity classification and the association of severity with neuropsychomotor development. Pediatric Radiology, 52, 941-950. https://doi.org/10.1007/s00247-022-05284-z
Farcy, S., Albert, A., Gressens, P., Baffet, A. D., & Ghouzzi, V. E. (2022). Cortical Organoids to Model Microcephaly. Cells, 11(14), 2135. https://doi.org/10.3390/cells11142135
Jgamadze, D., Lim, J. T., Zhang, Z., Harary, P. M., Germi, J., Mensah-Brown, K., Adam, C. D., Mirzakhalili, E., Singh, S., Gu, J. B., Blue, R., Dedhia, M., Fu, M., Jacob, F., Qian, X., Gagnon, K., Sergison, M., Fruchet, O., Rahaman, I., . . . Chen, H. I. (2023). Structural and functional integration of human forebrain organoids with the injured adult rat visual system. Cell Stem Cell, 30(2), 137-152.e7. https://doi.org/10.1016/j.stem.2023.01.004
Lancaster, M.A., Renner, M., Martin, C, Wenzel, D., Bicknell, L.S., Hurles, M.E., Homfrey, T., Penninger, J.M., Jackson, A.P., Knoblich, J.A. (2013). Cerebral organoids model human brain development and microcephaly. Nature 501, 373-379. https://doi.org/10.1038/nature12517
Mansour, A. A., Gonçalves, J. T., Bloyd, C. W., Li, H., Fernandes, S., Quang, D., Johnston, S., Parylak, S. L., Jin, X., & Gage, F. H. (2018). An in vivo model of functional and vascularized human brain organoids. Nature Biotechnology, 36(5), 432-441. https://doi.org/10.1038/nbt.4127
Rheinwatd, J. G., & Green, H. (1975). Seria cultivation of strains of human epidemal keratinocytes: The formation keratinizin colonies from single cell is. Cell, 6(3), 331-343. https://doi.org/10.1016/S0092-8674(75)80001-8
Samarasinghe, R. A., Miranda, O. A., Buth, J. E., Mitchell, S., Ferando, I., Watanabe, M., Allison, T. F., Kurdian, A., Fotion, N. N., Gandal, M. J., Golshani, P., Plath, K., Lowry, W. E., Parent, J. M., Mody, I., & Novitch, B. G. (2021). Identification of neural oscillations and epileptiform changes in human brain organoids. Nature Neuroscience, 24(10), 1488-1500. https://doi.org/10.1038/s41593-021-00906-5
Sato, T., Vries, R. G., Snippert, H. J., Barker, N., Stange, D. E., Van Es, J. H., Abo, A., Kujala, P., Peters, P. J., & Clevers, H. (2009). Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature, 459(7244), 262-265. https://doi.org/10.1038/nature07935
Wimmer, R. A., Leopoldi, A., Aichinger, M., Kerjaschki, D., & Penninger, J. M. (2019). Generation of blood vessel organoids from human pluripotent stem cells. Nature Protocols, 14(11), 3082-3100. https://doi.org/10.1038/s41596-019-0213-z
Yang, S., Hu, H., Kung, H., Zou, R., Dai, Y., Hu, Y., Wang, T., Lv, T., Yu, J., & Li, F. (2023). Organoids: The current status and biomedical applications. MedComm, 4(3), e274. https://doi.org/10.1002/mco2.274
Zhang, Y., Tang, C., He, Y., Zhang, Y., Li, Q., Zhang, T., Zhao, B., Tong, A., Zhong, Q., & Zhong, Z. (2024). Semaglutide ameliorates Alzheimer's disease and restores oxytocin in APP/PS1 mice and human brain organoid models. Biomedicine & Pharmacotherapy, 180, 117540. https://doi.org/10.1016/j.biopha.2024.117540


Comments